 
In the past, patrons of a cinema complex have spent an average.Using Table 8 below, determine the molar mass of sodium carbonate, Na2CO3. Table 7: Molar Mass of Calcium Chloride (CaCl2) Atomic Mass Multiply # in Contribution Formula Element Calcium (Ca) g/mol g/mol Chlorine (CI) g/mol g/mol Total = g/mol 3. Using Table 7 below, determine the molar mass of calcium chloride, CaCl2. 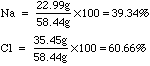
Balance the following chemical reaction: Na2CO3 (aq) +CaCl2 (aq) → CaCO3 (s)+NaCl (aq) Na₂CO₃ + Call, CaCO3 + 2NaCl 2. Balance the following chemical reaction: Na2CO3 (aq) +CaCl2 (aq) → CaCO3 (s)+NaCl (aq) Na₂CO₃ +.ġ. What volume of seawater contains 5.0 kg of Seawater contains approximately 3.5%NaCl by mass and has aĭensity of 1.02 g/mL. Seawater contains approximately 3.5%NaCl by mass and has a.Many grams of NaCl (1 molar mass = 58.44 g/mol) are required to The given amount of NaCl (molar mass 58.44g/mol )and final calculate the molarity of each aqueous solution with the given amount of NaCl (molar mass 58.44g/mol )and final volumeĬalculate the molarity of each aqueous solution with. 
Hydrogen chloride is prepared using the reaction: NaCl(aq) + H2SO4(aq) → _HCl(9) + _Na2SO4(aq) What is the limiting reagent if 4.00 g H2S04 (98.09) is reacted with 393 moles NaCl (58.44) You must balance the equation first. How many oxygens are in the molecular formula? Include the number only.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
